DNA Adduct Molecular Probes
A project funded by the European Research Council, supporting investigator-driven frontier research. An aim of ERC grants is to stimulate scientific excellence by supporting the very best, truly creative scientists to be adventurous and take risks in their research.
Elucidating the Diet-Cancer Connection at Chemical Resolution
Nitrosamines are potent carcinogens formed in processed foods, such as cured meats, beer, and cheese, and are components of tobacco smoke. They are bioactivated to electrophilic intermediates that react with DNA to form, amongst others, O6-alkyl-dG adducts that, if not corrected by repair enzymes can result cancer-causing genetic mutations.
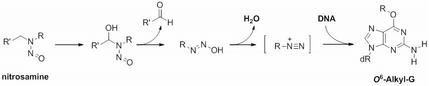
To understand the biological impacts of corresponding low-level bulky O6-alkyl-dG adducts, we have devised a new class of synthetic nucleoside probes for recognizing this type of DNA damage and are exploring strategies for implementing these novel molecules as mechanistic probes and the basis of new detection methods.
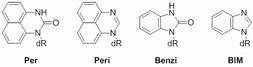
Our project “DNA Adduct Molecular Probes: Elucidating the Diet-Cancer Connection at Chemical Resolution,” which is funded by an ERC Grant, has the following aims:
- To characterize adduct:probe base-pairs in DNA duplexes
- To utilize nucleoside analogs in enzyme-mediated DNA synthesis reactions
- To develop DNA hybridization probes for sequence specific detection of bulky DNA adducts in cancer genes
Characterize Adduct:Probe Base-Pairs in DNA Duplexes
We have incorporated nucleoside analogs into oligonucleotide hybridization probes for detecting DNA adducts. These probes hybridize with complementary damaged DNA, such that the nucleoside analog P pairs with O6-alkyl-dG or non-damaged G. Discrimination between the adduct and the non-damaged strand can occur on the basis of DNA duplex stability or on differential fluorescence of the nucleoside analog:
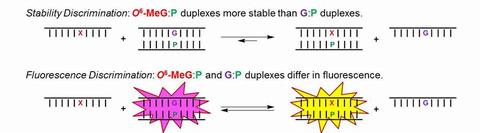
In this sub-project, we synthesize and incorporate nucleoside analogs (P) into oligonucleotides and assess fluorescence and other biophysical properties. By creating novel probe structures and targeting varying adduct structures, we can define changes in the properties of the DNA duplex on the basis of base pairing interactions.
Nucleoside Analogs in Enzyme-Mediated DNA Synthesis
DNA adducts alter the size and shape of DNA. During DNA replication, DNA adducts can cause mutations during polymerase-mediated translesion synthesis (TLS) or Post-lesion synthesis (PLS).
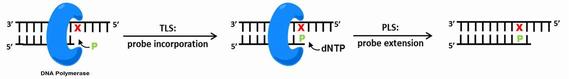
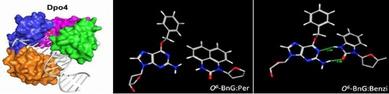
In this sub-project, we study DNA synthesis at and past base pairs consisting of DNA adducts opposite nucleoside analogs. We perform biochemical studies of polymerase-mediated synthesis using chemically modified templates, primers and triphosphates. Additionally, computational modeling enables us to envision the geometries of these pairs within the active site of the polymerase, such as the error-prone lesion bypass polymerase Dpo4:
DNA Hybridization Probes for Sequence-Specific DNA Adduct Detection
The synthetic nucleoside probes used in the mechanistic studies described above have potential applications for DNA damage detection and quantification. Toward this end, we create and study novel DNA-functionalized gold nanoparticles (Au NPs) and emissive oligonucleotides. A long-term goal is to develop a convenient and sensitive sequence-specific bioanalytical strategy for monitoring DNA damage.
In this sub-project, we prepare and characterize by spectroscopic techniques functionalized oligonucleotides and AuNPs. We design and test novel biosensing strategies for chemically modified DNA, and aim to exploit changes in optical properties for quantitative analysis.
